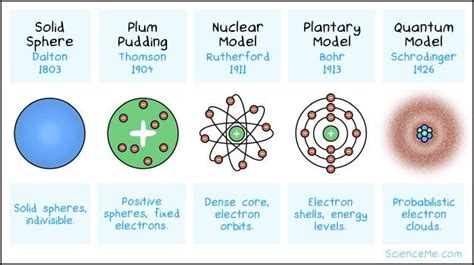
The concept of atomic models has been a cornerstone of chemistry and physics for centuries, with various scientists contributing to our understanding of the structure and behavior of atoms. From the early days of Dalton’s atomic theory to the modern quantum mechanical models, the development of atomic models has been a gradual process, shaped by experimental evidence and theoretical insights.
One of the earliest and most influential atomic models was proposed by John Dalton in the early 19th century. Dalton’s model, which is often referred to as the “billiard ball” model, posits that atoms are indivisible, spherical particles that retain their properties regardless of the compound they form. While Dalton’s model was groundbreaking for its time, it had several limitations, including its inability to explain the behavior of atoms in chemical reactions.
The discovery of subatomic particles, such as electrons, protons, and neutrons, revolutionized our understanding of atomic structure. The “plum pudding” model, proposed by J.J. Thomson, suggested that atoms consist of a positively charged sphere with negatively charged electrons embedded within. However, this model was later disproven by Ernest Rutherford’s famous gold foil experiment, which demonstrated that atoms have a small, dense nucleus surrounded by electrons.
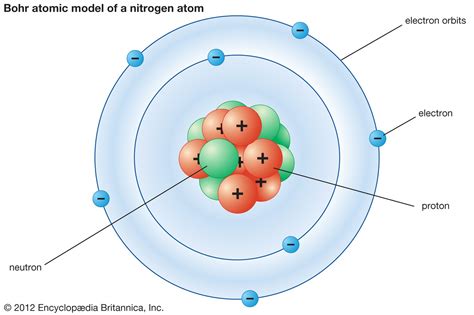
Rutherford’s “planetary” model, which placed the nucleus at the center of the atom with electrons orbiting around it, marked a significant improvement over earlier models. However, it was not without its limitations, as it failed to explain the behavior of electrons in terms of energy and stability. The introduction of quantum mechanics by Niels Bohr and Louis de Broglie provided a major breakthrough, as it introduced the concept of energy levels and electron spin.
The quantum mechanical model of the atom, also known as the “electron cloud” model, is the most widely accepted model today. This model describes the behavior of electrons in terms of probability distributions, rather than definite orbits, and takes into account the principles of wave-particle duality and uncertainty. The electron cloud model is supported by a vast amount of experimental evidence, including spectroscopic data and electron microscopy images.
Despite its success, the quantum mechanical model is not without its limitations. The model assumes that the nucleus is a fixed, point-like object, which is not entirely accurate. Additionally, the model does not account for the behavior of atoms in extreme environments, such as high temperatures and pressures. To address these limitations, researchers have developed more advanced models, such as the relativistic quantum mechanics and quantum field theory.
In conclusion, the development of atomic models has been a rich and complex process, shaped by the contributions of many scientists over the centuries. From the early “billiard ball” model to the modern quantum mechanical models, our understanding of atomic structure and behavior has undergone significant transformations. While there is still much to be learned, the current models provide a solid foundation for understanding the behavior of atoms and their role in the physical world.
Historical Evolution of Atomic Models
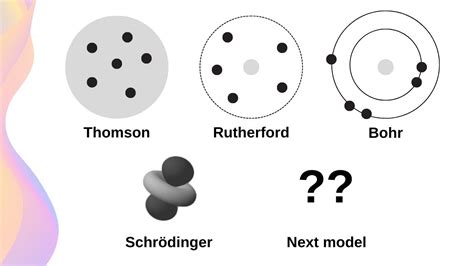
The historical evolution of atomic models is a fascinating story that reflects the gradual development of scientific understanding. From the early Greek philosophers to the modern quantum mechanical models, the concept of the atom has undergone significant transformations.
One of the earliest recorded discussions of the atom was by the Greek philosopher Democritus, who proposed that matter is composed of indivisible, eternal atoms. However, this idea was not widely accepted until the 19th century, when John Dalton developed his atomic theory.
The discovery of subatomic particles, such as electrons and protons, marked a significant turning point in the development of atomic models. The “plum pudding” model, proposed by J.J. Thomson, was one of the first attempts to incorporate these new discoveries into a comprehensive model of the atom.
However, it was not until the development of quantum mechanics that the modern understanding of the atom began to take shape. The introduction of wave-particle duality, uncertainty principle, and electron spin provided a new framework for understanding the behavior of atoms and molecules.
- Dalton's atomic theory: proposed that atoms are indivisible, spherical particles
- Discovery of subatomic particles: electrons, protons, and neutrons
- Rutherford's planetary model: placed the nucleus at the center of the atom with electrons orbiting around it
- Introduction of quantum mechanics: provided a new framework for understanding the behavior of atoms and molecules
- Development of the quantum mechanical model: describes the behavior of electrons in terms of probability distributions
Technical Breakdown of Quantum Mechanical Model
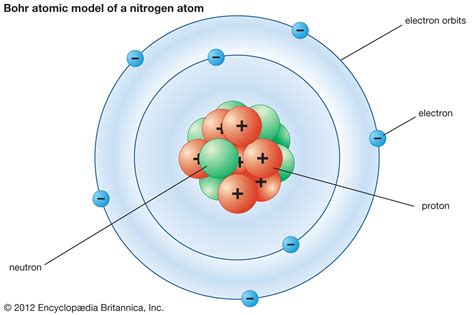
The quantum mechanical model of the atom is a complex and highly mathematical framework that describes the behavior of electrons in terms of probability distributions. At its core, the model is based on the principles of wave-particle duality, uncertainty principle, and electron spin.
One of the key features of the quantum mechanical model is the concept of energy levels. Electrons in an atom occupy specific energy levels, or orbitals, which are characterized by a set of quantum numbers. These energy levels are not fixed, but rather are influenced by the presence of other electrons and the nucleus.
The behavior of electrons in an atom is also influenced by the principles of wave-particle duality and uncertainty principle. According to wave-particle duality, electrons can exhibit both wave-like and particle-like behavior, depending on the experimental conditions. The uncertainty principle, on the other hand, states that it is impossible to know certain properties of an electron, such as its position and momentum, simultaneously with infinite precision.
- Provides a highly accurate description of the behavior of atoms and molecules
- Allows for the calculation of energy levels and other properties of atoms and molecules
- Provides a framework for understanding the behavior of electrons in different environments
- Is highly mathematical and requires a significant amount of computational power
- Assumes that the nucleus is a fixed, point-like object, which is not entirely accurate
- Does not account for the behavior of atoms in extreme environments, such as high temperatures and pressures
What is the main difference between the "plum pudding" model and the quantum mechanical model of the atom?
+
The main difference between the "plum pudding" model and the quantum mechanical model is the way they describe the behavior of electrons. The "plum pudding" model suggests that electrons are embedded within a positively charged sphere, while the quantum mechanical model describes electrons in terms of probability distributions.
What is the significance of energy levels in the quantum mechanical model of the atom?
+
Energy levels are a key feature of the quantum mechanical model, as they describe the specific energies that electrons can occupy in an atom. These energy levels are influenced by the presence of other electrons and the nucleus, and they play a critical role in determining the chemical properties of an atom.
How does the quantum mechanical model account for the behavior of electrons in different environments?

+
The quantum mechanical model accounts for the behavior of electrons in different environments by incorporating the principles of wave-particle duality and uncertainty principle. These principles allow for the calculation of energy levels and other properties of atoms and molecules in different environments, such as high temperatures and pressures.
In conclusion, the quantum mechanical model of the atom is a highly successful framework that describes the behavior of electrons in terms of probability distributions. While it has several limitations, the model provides a solid foundation for understanding the behavior of atoms and molecules, and it has been widely used in a variety of fields, including chemistry, physics, and materials science.