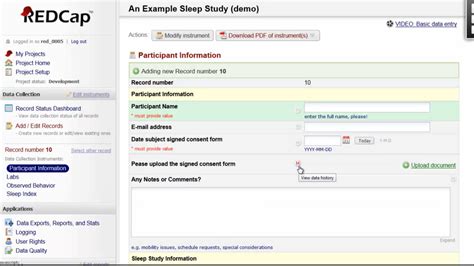
Introduction to Redcap Clinical Research Management Solution

Redcap is a popular clinical research management solution that enables researchers to efficiently collect, manage, and analyze data. Developed by Vanderbilt University, Redcap has become a widely used platform in the clinical research community due to its flexibility, scalability, and user-friendly interface. In this blog post, we will delve into the features, benefits, and applications of Redcap, highlighting its importance in streamlining clinical research processes.
Key Features of Redcap

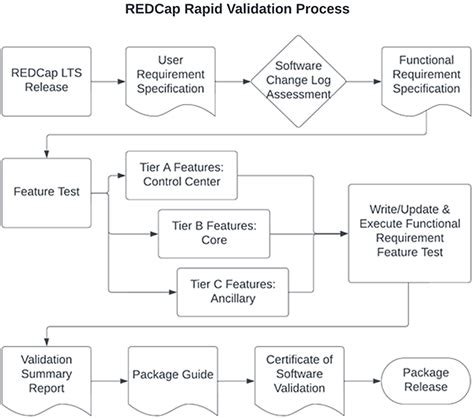
Redcap offers a range of features that cater to the diverse needs of clinical researchers. Some of the key features include: * Data Collection: Redcap allows researchers to create custom surveys, forms, and databases to collect data from various sources, including patients, clinicians, and laboratories. * Data Management: The platform provides a secure and centralized repository for storing and managing data, with features such as data validation, data normalization, and data encryption. * Data Analysis: Redcap offers built-in tools for data analysis, including statistical analysis, data visualization, and reporting. * Collaboration: The platform enables real-time collaboration among researchers, allowing them to share data, track changes, and communicate with each other. * Security: Redcap ensures the security and integrity of data through features such as user authentication, access control, and audit trails.
Benefits of Using Redcap
The use of Redcap in clinical research offers several benefits, including: * Improved Data Quality: Redcap’s data validation and normalization features help ensure that data is accurate, complete, and consistent. * Increased Efficiency: The platform automates many tasks, such as data collection and data entry, freeing up researchers to focus on higher-level tasks. * Enhanced Collaboration: Redcap’s collaboration features facilitate communication and coordination among researchers, promoting a more efficient and effective research process. * Reduced Costs: By streamlining data collection and management, Redcap helps reduce the costs associated with clinical research. * Compliance: Redcap is designed to meet regulatory requirements, such as HIPAA and FDA guidelines, ensuring that research is conducted in a compliant and ethical manner.
Applications of Redcap in Clinical Research
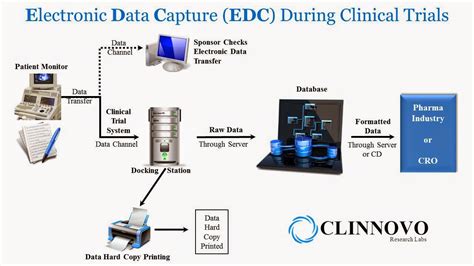
Redcap has a wide range of applications in clinical research, including: * Clinical Trials: Redcap is used to manage data from clinical trials, including patient enrollment, randomization, and outcome assessment. * Observational Studies: The platform is used to collect and manage data from observational studies, such as cohort studies and case-control studies. * Registry Studies: Redcap is used to manage data from registry studies, including disease registries and patient registries. * Quality Improvement Initiatives: The platform is used to collect and analyze data for quality improvement initiatives, such as patient safety and quality of care.
| Application | Description |
|---|---|
| Clinical Trials | Management of data from clinical trials, including patient enrollment and outcome assessment |
| Observational Studies | Collection and management of data from observational studies, such as cohort studies and case-control studies |
| Registry Studies | Management of data from registry studies, including disease registries and patient registries |
| Quality Improvement Initiatives | Collection and analysis of data for quality improvement initiatives, such as patient safety and quality of care |

Best Practices for Implementing Redcap

To ensure successful implementation of Redcap, researchers should follow best practices, including: * Define Clear Goals and Objectives: Clearly define the goals and objectives of the research project, including the type of data to be collected and the desired outcomes. * Develop a Data Management Plan: Develop a data management plan that outlines the procedures for collecting, storing, and managing data. * Train Users: Provide training to users on the use of Redcap, including data entry, data management, and data analysis. * Monitor and Evaluate: Continuously monitor and evaluate the use of Redcap, identifying areas for improvement and implementing changes as needed.
📝 Note: Regular training and support are essential for ensuring that users are proficient in the use of Redcap and that data is collected and managed accurately and efficiently.
Future Directions for Redcap
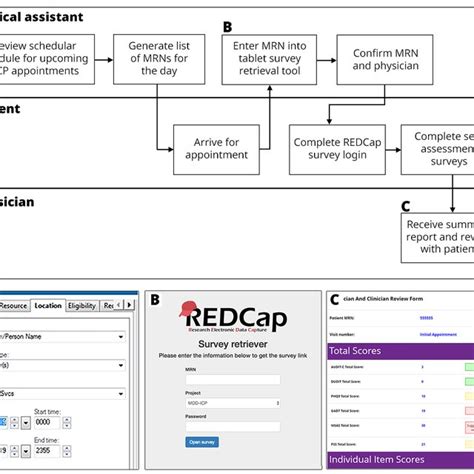
As clinical research continues to evolve, Redcap is likely to play an increasingly important role in streamlining research processes. Future directions for Redcap may include: * Integration with Electronic Health Records: Integration of Redcap with electronic health records (EHRs) to enable seamless data exchange and reduce data duplication. * Artificial Intelligence and Machine Learning: Incorporation of artificial intelligence and machine learning algorithms to enhance data analysis and identify patterns and trends. * Mobile Device Support: Development of mobile device support to enable researchers to collect and manage data in real-time, using mobile devices such as smartphones and tablets.
In summary, Redcap is a powerful clinical research management solution that offers a range of features and benefits to support the efficient collection, management, and analysis of data. By following best practices and staying up-to-date with the latest developments, researchers can maximize the potential of Redcap and advance the field of clinical research.
What is Redcap?
+
Redcap is a clinical research management solution that enables researchers to efficiently collect, manage, and analyze data.
What are the key features of Redcap?
+
The key features of Redcap include data collection, data management, data analysis, collaboration, and security.
What are the benefits of using Redcap?
+
The benefits of using Redcap include improved data quality, increased efficiency, enhanced collaboration, reduced costs, and compliance with regulatory requirements.