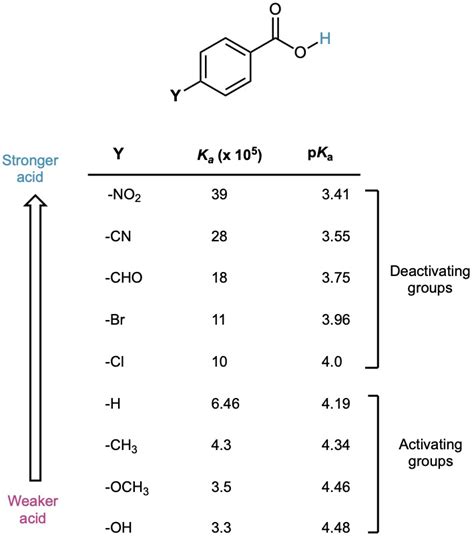
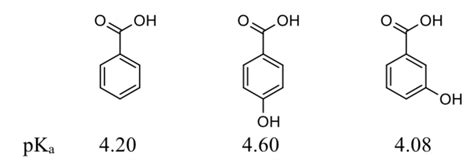
Understanding the pKa value of benzoic acid is crucial in various chemical and pharmaceutical applications, as it influences the compound’s reactivity, solubility, and interaction with other molecules. Benzoic acid, a weak organic acid, is widely used as a food preservative, in the manufacture of dyes, and as an intermediate in the synthesis of pharmaceuticals and other organic compounds. The pKa value, which is the negative logarithm of the acid dissociation constant (Ka), is a quantitative measure of the strength of an acid in solution. For benzoic acid, the pKa value is approximately 4.19 at 25°C.
To delve into the significance of this value, it’s essential to grasp the concept of acid dissociation. When benzoic acid (C6H5COOH) is dissolved in water, it partially dissociates into its conjugate base, benzoate (C6H5COO-), and a proton (H+), according to the equation: C6H5COOH ⇌ C6H5COO- + H+. The pKa value of 4.19 indicates that at a pH of 4.19, the concentrations of benzoic acid and its conjugate base are equal. This equilibrium constant is vital for predicting the behavior of benzoic acid in different environments.
Theoretical Background
The acid dissociation constant (Ka) is defined by the equation Ka = [C6H5COO-][H+]/[C6H5COOH], where [C6H5COO-], [H+], and [C6H5COOH] are the concentrations of benzoate, proton, and benzoic acid, respectively. The pKa is then calculated as pKa = -log10(Ka). The pKa value provides a direct measure of an acid’s strength; lower pKa values indicate stronger acids, as they more readily donate a proton.
Practical Applications
Pharmaceuticals: Understanding the pKa of benzoic acid is crucial in drug development, especially for drugs that are weak acids or bases. The pKa influences how a drug is absorbed, distributed, metabolized, and excreted (ADME) in the body. For instance, the ionization state of a drug at different pH values in the body can affect its ability to cross biological membranes.
Food Preservation: Benzoic acid’s antimicrobial properties make it an effective preservative in foods and beverages. Its pKa value helps in understanding its effectiveness at different pH levels. The undissociated form of benzoic acid is more lipid-soluble and can penetrate microbial cell membranes more easily, thereby inhibiting the growth of yeast, mold, and bacteria.
Synthetic Chemistry: In organic synthesis, the pKa of benzoic acid is a critical parameter for selecting appropriate reaction conditions, such as the choice of solvent, catalyst, or base, to facilitate specific transformations. For example, the deprotonation of benzoic acid to form its conjugate base can be a crucial step in certain synthetic pathways.
Impact of Temperature and Solvent

The pKa value of benzoic acid is not constant and can be influenced by factors such as temperature and the solvent used. An increase in temperature generally decreases the pKa of an acid, indicating an increase in the acid’s dissociation constant. Similarly, the choice of solvent can significantly affect the pKa; solvents with higher dielectric constants can stabilize the charged species (in this case, benzoate and proton) more effectively, leading to an increase in the dissociation constant and a corresponding decrease in the pKa value.
Experimental Determination
The pKa of benzoic acid can be experimentally determined through various methods, including potentiometric titration, spectroscopic methods, and chromatographic techniques. Potentiometric titration involves the gradual addition of a strong base to a solution of benzoic acid while monitoring the pH. The point of inflection in the resulting titration curve corresponds to the pKa of benzoic acid.
Conclusion

In conclusion, the pKa value of benzoic acid is a fundamental parameter that underlies its chemical properties and applications. Understanding this value and its implications is essential for chemists, pharmacists, and food scientists, among others, to predict and manipulate the behavior of benzoic acid in various contexts. Whether it’s designing new pharmaceuticals, optimizing food preservation strategies, or synthesizing complex organic molecules, knowledge of the pKa of benzoic acid is indispensable.
What is the significance of the pKa value of benzoic acid in pharmaceutical applications?
+
The pKa value of benzoic acid is significant in pharmaceutical applications because it influences the absorption, distribution, metabolism, and excretion (ADME) of drugs. The ionization state of a drug, as determined by its pKa and the pH of the environment, can affect its ability to cross biological membranes, thereby impacting its efficacy and toxicity.
How does the pKa of benzoic acid affect its use as a food preservative?
+
The pKa of benzoic acid affects its use as a food preservative by influencing its effectiveness at different pH levels. The undissociated form of benzoic acid, which predominates at lower pH values, is more effective at inhibiting the growth of microorganisms due to its higher lipid solubility and ability to penetrate microbial cell membranes.
What factors can influence the pKa value of benzoic acid?
+
The pKa value of benzoic acid can be influenced by factors such as temperature and the solvent used. An increase in temperature or the use of a solvent with a higher dielectric constant can decrease the pKa value, indicating an increase in the acid’s dissociation constant.