Introduction to Benzoic Acid PKA

Benzoic acid is a compound that has been widely used in various industries, including food, pharmaceutical, and cosmetic. One of the key properties of benzoic acid is its acid dissociation constant, also known as pKa. The pKa value of benzoic acid is a crucial factor in determining its behavior and applications. In this article, we will discuss 5 tips about benzoic acid pKa and its significance.
Tip 1: Understanding pKa Value
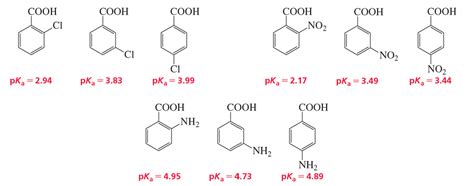
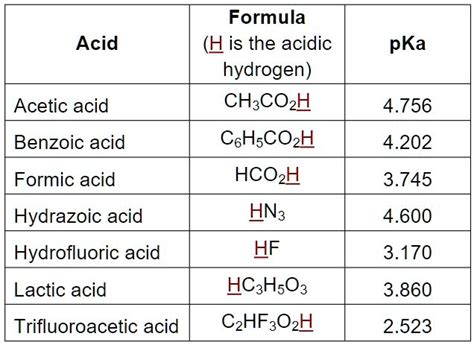
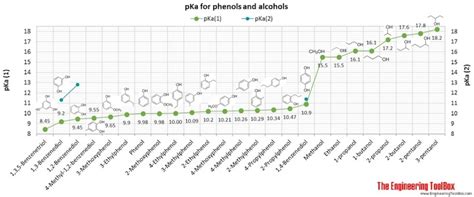
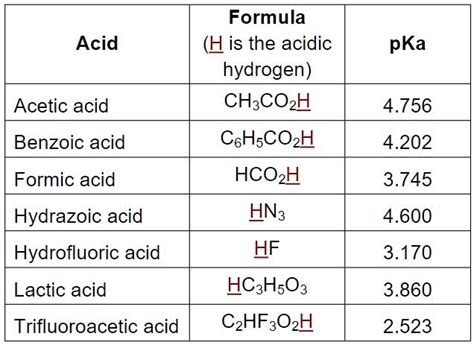
The pKa value of benzoic acid is approximately 4.2, which indicates that it is a weak acid. This means that benzoic acid only partially dissociates in water, releasing a proton (H+ ion) and forming a benzoate ion. The pKa value is a measure of the strength of an acid, with lower values indicating stronger acids and higher values indicating weaker acids. Understanding the pKa value of benzoic acid is essential in predicting its behavior in different environments.
Tip 2: Factors Affecting pKa Value
Several factors can affect the pKa value of benzoic acid, including: * Temperature: Changes in temperature can influence the pKa value of benzoic acid. Generally, an increase in temperature leads to a decrease in pKa value. * Solvent: The pKa value of benzoic acid can vary depending on the solvent used. For example, the pKa value of benzoic acid in water is different from that in ethanol. * Concentration: The concentration of benzoic acid can also impact its pKa value. At higher concentrations, the pKa value may decrease due to increased ionization.
Tip 3: Applications of Benzoic Acid pKa
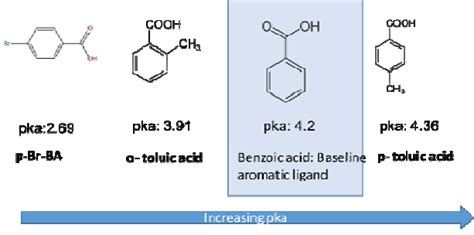
The pKa value of benzoic acid has significant implications for its applications. Some of the key uses of benzoic acid include: * Food preservation: Benzoic acid is used as a preservative in food products, such as jams, jellies, and beverages. Its pKa value helps to determine its effectiveness in inhibiting the growth of microorganisms. * Pharmaceuticals: Benzoic acid is used in the production of various pharmaceuticals, including antifungal and antibacterial agents. Its pKa value is crucial in determining its bioavailability and efficacy. * Cosmetics: Benzoic acid is used in the production of cosmetics, such as skin care products and shampoos. Its pKa value helps to determine its stability and effectiveness in these applications.
Tip 4: Measuring pKa Value
The pKa value of benzoic acid can be measured using various methods, including: * Potentiometric titration: This method involves titrating a solution of benzoic acid with a strong base, such as sodium hydroxide, and measuring the pH at different points. * Spectrophotometry: This method involves measuring the absorbance of a solution of benzoic acid at different wavelengths and calculating the pKa value from the data. * Chromatography: This method involves separating a mixture of benzoic acid and its conjugate base using chromatography and measuring the pKa value from the retention times.
Tip 5: Importance of pKa Value in Chemical Reactions
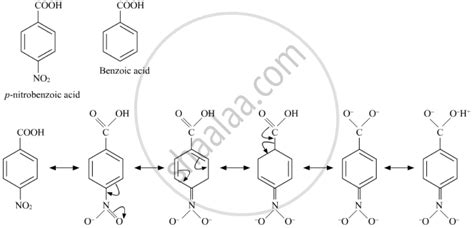
The pKa value of benzoic acid is critical in determining its behavior in chemical reactions. For example: * Acid-base reactions: The pKa value of benzoic acid determines its ability to donate or accept protons in acid-base reactions. * Complexation reactions: The pKa value of benzoic acid can influence its ability to form complexes with metal ions. * Hydrolysis reactions: The pKa value of benzoic acid can affect its susceptibility to hydrolysis reactions.
🔍 Note: Understanding the pKa value of benzoic acid is essential in predicting its behavior and applications in various industries.
In summary, the pKa value of benzoic acid is a crucial factor in determining its behavior and applications. By understanding the factors that affect the pKa value, measuring it accurately, and considering its importance in chemical reactions, we can harness the full potential of benzoic acid in various industries.
What is the pKa value of benzoic acid?
+
The pKa value of benzoic acid is approximately 4.2.
What factors affect the pKa value of benzoic acid?
+
Temperature, solvent, and concentration can affect the pKa value of benzoic acid.
What are the applications of benzoic acid?
+
Benzoic acid is used in food preservation, pharmaceuticals, and cosmetics.